A Lewis structure of aspirin without the nonbonding electrons is shown in the figure below. Taking into account the nonbonding electrons, identify the hybridization of the atomic orbitals for the following atoms: O1, O2, and O3. Identify the bond angles around C1, C2, and O3. 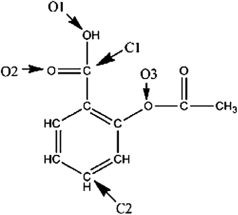
Correct Answer:
Verified
View Answer
Unlock this answer now
Get Access to more Verified Answers free of charge
Q162: Identify the molecular geometry of the nitrate
Q163: In the following molecules, what orbitals are
Q164: Describe the valence bond picture of
Q165: The amide structure is the fundamental linking
Q166: Draw the correct orientation of the dipole
Q168: Identify the electron pair and molecular geometries
Q169: Identify the hybridization of the atomic orbitals
Q170: Carbonyl dihalides (COX2 with X = I,
Q171: In 1995 a Japanese cult attacked the
Q172: Predict the bond angles at the three
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents