The following reaction represents the self-ionization of ammonia (NH3).
NH3(aq)+ NH3(aq) 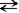 NH2-(aq)+ NH4+(aq)
NH2-(aq)+ NH4+(aq)
amide ammonium
Fill in the blanks with the appropriate terms from those listed below.
ammonia
ammonium
amide
-In the reverse reaction, __________________________ion functions as an acid.
Correct Answer:
Verified
Q65: If you dissolve 0.10 mol of a
Q66: Consider the following three buffer reactions.
Buffer 1:
Q67: Arterial blood gases (ABGs) were drawn on
Q68: Joshua has had high blood pressure for
Q69: Consider the following three buffer reactions.
Buffer 1:
Q71: Consider the following three buffer reactions.
Buffer 1:
Q72: What is the pH of a solution
Q73: Examine the structure given below 
Q74: The buffer solution in the flask was
Q75: In a solution of phosphoric acid all
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents