Upon electrification, hydrogen produces a line spectrum with the following lines in the visible region of the electromagnetic spectrum. If the light emitted corresponds to transitions from the third (n = 3) , fourth (n = 4) , fifth (n = 5) , or sixth (n = 6) energy level down to the second (n = 2) , which transition corresponds to the emission of light with highest energy? 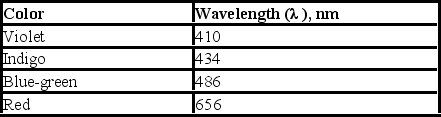
A) n = 2 → n = 3
B) n = 2 → n = 6
C) n = 3 → n = 2
D) n = 4 → n = 2
E) n = 6 → n = 3
Correct Answer:
Verified
Q16: A laser used in DVD players has
Q17: Rank the following types of electromagnetic radiation
Q18: Which of the following statements regarding the
Q19: The wavelength of the blue light given
Q20: The wavelength of the blue light given
Q22: Which of the images represents a p
Q23: Upon electrification, hydrogen produces a characteristic line
Q24: The green line observed in the line
Q25: Which of the following statements regarding orbitals
Q26: The green line observed in the line
Unlock this Answer For Free Now!
View this answer and more for free by performing one of the following actions

Scan the QR code to install the App and get 2 free unlocks

Unlock quizzes for free by uploading documents